
Đang tải...

Đang tải...

Photo: Pixabay/CC0 Public Domain
Many electric vehicle owners worry about how efficient their batteries will be in very cold weather. Now a new battery chemistry may have solved that problem.
In the current lithium-ion battery, the main problem lies in the liquid electrolyte. The key component of this battery transfers charge carriers called ions between the battery's two electrodes, causing the battery to charge and discharge. But the liquid starts to freeze at sub-zero temperatures. This situation severely limits the efficiency of electric vehicle charging in cold regions and cold seasons.
To solve that problem, a team of scientists from the US Department of Energy's (DOE) Argonne and Lawrence Berkeley National Laboratory has developed a fluorine-containing electrolyte that performs well even at high temperatures. below 0.
The research appeared in Advanced Energy Materials.
"Our team not only found an antifreeze electrolyte whose charging efficiency does not drop at minus 4 degrees Fahrenheit, but we also discovered, at the atomic level, what makes it so efficient. ", said Zhengcheng "John" Zhang, a senior chemist and team leader in Argonne's Chemical Engineering and Science division.
This low-temperature electrolyte shows promise for batteries in electric vehicles, as well as in energy storage for the grid and consumer electronics such as computers and phones.
In today's lithium-ion batteries, the electrolyte is a mixture of a widely available salt (lithium hexafluorophosphate) and a carbonate solvent such as ethylene carbonate. Solvents dissolve salts to form liquids.
When the battery is charged, the liquid electrolyte transfers lithium ions from the cathode (a lithium-containing oxide) to the anode (graphite). These ions move out of the cathode, then through the electrolyte on their way into the anode. While transported through the electrolyte, they are located at the center of clusters of four or five solvent molecules.
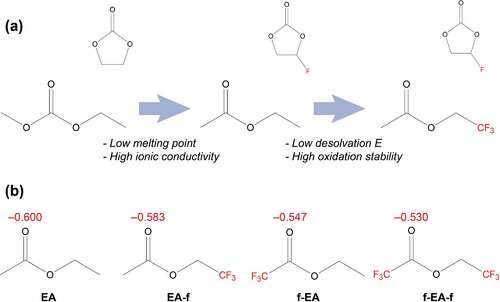
a) Schematic diagram of solvent conversion from carbonate to fluorinated ester. b) Atomic charge analysis of the carbonyl groups in EA, EA-f, f-EA and f-EA-f.
During the initial few charges, these clusters attack the anode surface and form a protective layer called solid electrolyte intercalation. Once formed, this layer acts as a filter. It only allows lithium ions to pass through the layer while blocking solvent molecules. In this way, the anode can store lithium atoms in the structure of charged graphite. During discharge, electrochemical reactions release electrons from the lithium creating electricity that can power a vehicle.
The problem is that at cold temperatures the electrolyte with the carbonate solvent starts to freeze. As a result, it loses its ability to transport lithium ions to the anode when charged. This is because the lithium ions are very tightly bound in the solvent clusters. Therefore, these ions require much higher energy to evacuate their clusters and penetrate the interface layer than at room temperature. For that reason, scientists searched for a better solvent.
The team investigated several fluorinated solvents. They were able to identify the component with the lowest energy barrier to release lithium ions from clusters at sub-zero temperatures. They also determined at the atomic scale why that particular ingredient worked so well. It depends on the position of the fluorine atoms in each solvent molecule and their number.
In testing with lab cells, the team's fluorinated electrolyte retained its ability to store energy—stable for 400 discharge cycles at minus 4 F. Even at temperatures below <> degrees That is, the capacity is equivalent to a cell with a common carbonate-based electrolyte at room temperature.
"Our study thus demonstrated how to tune the atomic structure of an electrolyte solvent to design new electrolytes for sub-zero temperatures," said Zhang.
Antifreeze electrolyte has bonus properties. It is much safer than currently used carbonate-based electrolytes, as it will not catch fire.
"We are patenting a safer and lower-temperature electrolyte and are currently looking for an industry partner to adapt it to one of their designs for lithium-ion batteries. ", said Zhang.
In addition to John Zhang, Argonne's authors are Dong-Joo Yoo, Qian Liu, and Minkyu Kim. The Berkeley Lab authors are Orion Cohen and Kristin Persson.